Inflammatory Skin Disease Models
Validated In Vivo Models and Expert Services
for Dermatology Drug Development

Accelerating Dermatology Drug Discovery
Inflammatory skin diseases such as atopic dermatitis, psoriasis, irritant contact dermatitis, and delayed-type hypersensitivity are complex, immune-mediated conditions that present major challenges in both clinical management and preclinical research. At Inotiv, we help biopharmaceutical partners accelerate dermatology drug discovery and development through a portfolio of validated in vivo skin inflammation models that closely replicate human disease.
Our in vivo dermatology models are optimized to evaluate the efficacy, mechanism of action, and safety of therapies across a broad range of inflammatory skin diseases. We offer customized study designs and comprehensive research support—including clinical scoring, histopathology, immune profiling, and biomarker analysis—to help advance your candidate with speed and scientific rigor. Our studies deliver robust, translatable data that support confident, data-driven decision-making in preclinical development.
In Vivo Models for Inflammatory Skin Diseases
Our portfolio of validated in vivo models supports translational research across a range of inflammatory dermatologic indications. Each model replicates key aspects of human disease, offering measurable clinical and histopathological endpoints to assess disease progression and therapeutic response.
DELAYED-TYPE HYPERSENSITIVITY (DTH)
Delayed-type hypersensitivity (DTH) is a T cell–mediated immune response that occurs 24–72 hours after antigen exposure and plays a central role in skin inflammation, autoimmune pathology, and host defense against intracellular pathogens. Driven by CD4+ and CD8+ T cells, DTH responses involve the release of IFN-γ and TNF-α, which promote macrophage recruitment, tissue swelling, and localized immune infiltration.
Chemically Induced Contact DTH Models
Our chemically induced contact DTH models provide reliable in vivo platforms for evaluating the efficacy of therapeutic compounds, using topical application of sensitizing agents to induce T cell-mediated skin inflammation.
- Acute Oxazolone-Induced DTH Mouse and Rat Model
- 2,4-Dinitrofluorobenzene (DNFB)-Induced DTH Mouse and Rat Model
- Fluorescein Isothiocyanate (FITC)-Induced DTH Mouse Model
Protein-Induced DTH Models
Our protein-induced DTH models offer flexible in vivo platforms for assessing antigen-specific immune responses, with protein antigens administered via injection into the ear, footpad, or knee joint depending on study design and client objectives.
- Keyhole Limpet Hemocyanin (KLH)-Induced DTH Mouse Model
- Methylated Bovine Serum Albumin (mBSA)-Induced DTH Mouse Model
- KLH-Induced DTH Mouse Model
- Ovalbumin-Induced DTH Mouse Model
- Gliadin-Induced DTH Mouse Model
Example Data
 Mice were sensitized on the abdomen (Day 0) and challenged on the right ear with oxazolone (Day 7). Ear thickness was measured pre-challenge (Day 7) and 24 hours post-challenge (Day 8). Animals received twice-daily water (red circles) or dexamethasone (orange triangles) from Day 0–8. A challenge-only group (grey boxes) received oxazolone on Day 7 only. Data represent mean ± SE ear caliper difference (right minus left).
Mice were sensitized on the abdomen (Day 0) and challenged on the right ear with oxazolone (Day 7). Ear thickness was measured pre-challenge (Day 7) and 24 hours post-challenge (Day 8). Animals received twice-daily water (red circles) or dexamethasone (orange triangles) from Day 0–8. A challenge-only group (grey boxes) received oxazolone on Day 7 only. Data represent mean ± SE ear caliper difference (right minus left).
 Mice were sensitized with subcutaneous administration of KLH emulsified in Freund’s complete adjuvant (Day 0) and challenged with KLH injected into each ear pinna (Day 7). Ear thickness was measured pre-challenge (Day 7) and daily through Day 10. Animals received daily vehicle (red circles) or cyclosporine A (CSA; orange triangles) from Days 0–9. A challenge-only group (grey boxes) received KLH on Day 7 only. Data represent mean ± SE ear caliper measurements.
Mice were sensitized with subcutaneous administration of KLH emulsified in Freund’s complete adjuvant (Day 0) and challenged with KLH injected into each ear pinna (Day 7). Ear thickness was measured pre-challenge (Day 7) and daily through Day 10. Animals received daily vehicle (red circles) or cyclosporine A (CSA; orange triangles) from Days 0–9. A challenge-only group (grey boxes) received KLH on Day 7 only. Data represent mean ± SE ear caliper measurements.
 Mice were sensitized on Day 0 with ovalbumin or gliadin emulsified in Freund’s complete adjuvant and challenged on Day 13 with the corresponding antigen injected into the left footpad. Animals received twice-daily vehicle or CSA from Days 0-13. Ankle thickness was measured pre-challenge (Day 13) and 24 hours post-challenge (Day 14) for vehicle + ovalbumin (orange circles), CSA + ovalbumin (light grey boxes), vehicle + gliadin (dark grey boxes) and CSA + gliadin (red circles) groups. IgG-treated animals (orange triangles) served as negative controls. Data represent mean ± SE ankle caliper measurements (mm).
Mice were sensitized on Day 0 with ovalbumin or gliadin emulsified in Freund’s complete adjuvant and challenged on Day 13 with the corresponding antigen injected into the left footpad. Animals received twice-daily vehicle or CSA from Days 0-13. Ankle thickness was measured pre-challenge (Day 13) and 24 hours post-challenge (Day 14) for vehicle + ovalbumin (orange circles), CSA + ovalbumin (light grey boxes), vehicle + gliadin (dark grey boxes) and CSA + gliadin (red circles) groups. IgG-treated animals (orange triangles) served as negative controls. Data represent mean ± SE ankle caliper measurements (mm).
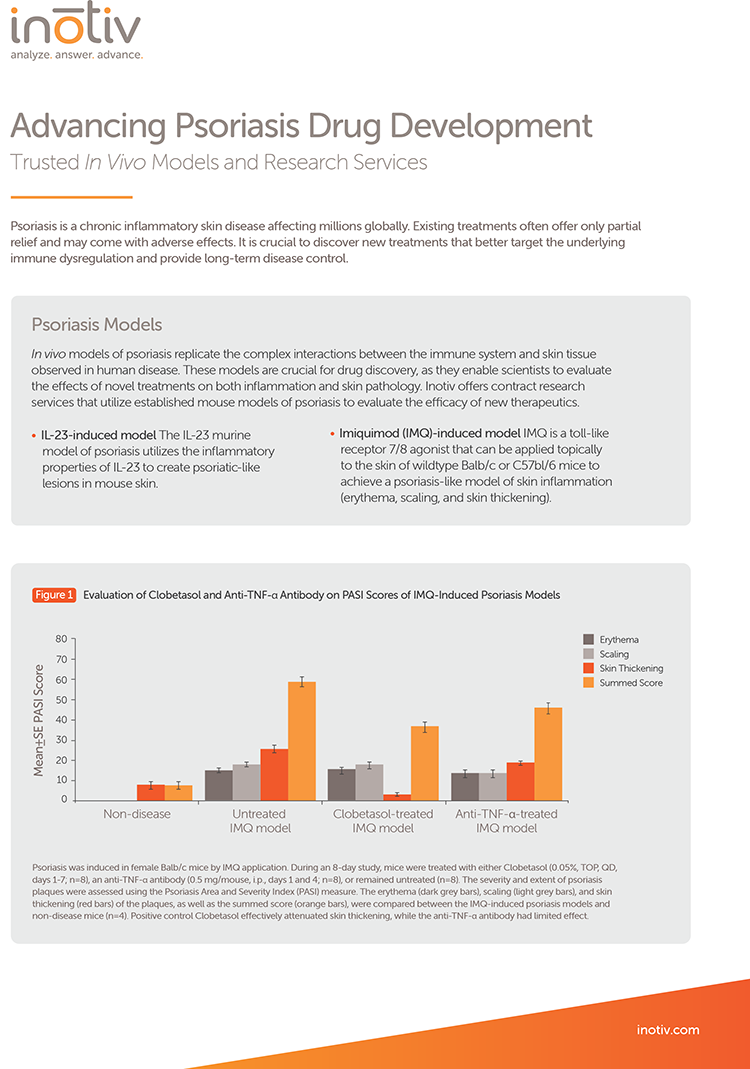
Animals were sensitized with mBSA in Freund’s complete adjuvant on Days 0 and 7 (base of tail) and challenged with mBSA administered into the right tibiotarsal joint on Day 21. Animals received vehicle or a JAK inhibitor daily from Days 0–34. (A) Ankle diameter was measured on Days 21–35 for vehicle-treated (orange triangles), JAK inhibitor–treated (red circles), and sensitized-only control animals (grey boxes). Data represent mean ± SE ankle diameter difference between treated and control ankles. (B) Mean histopathology scores for inflammation (dark grey bars), pannus formation (yellow bars), cartilage damage (light grey bars), bone resorption (red bars), and periosteal bone formation (orange bars) in ankle tissue.
ATOPIC DERMATITIS
Atopic dermatitis is a chronic, relapsing inflammatory skin disease characterized by eczema, intense pruritus, and epidermal barrier dysfunction. It is driven by a dominant Th2 immune response involving elevated IL-4, IL-13, IL-5, and IgE levels. In more severe or chronic forms, Th1, Th2, and Th17 cells may also contribute to inflammation and disease persistence. Our hapten-based atopic dermatitis models offer precise experimental control and high reproducibility, making them ideal for studying immune mechanisms, barrier dysfunction, and chronic inflammation.
Key Models
- Chronic Oxazolone-Induced Atopic Dermatitis Mouse Model
- Chronic FITC-Induced Atopic Dermatitis Mouse Model
- MC903 (Calcipotriol)-Induced Atopic Dermatitis Mouse Model
Example Data
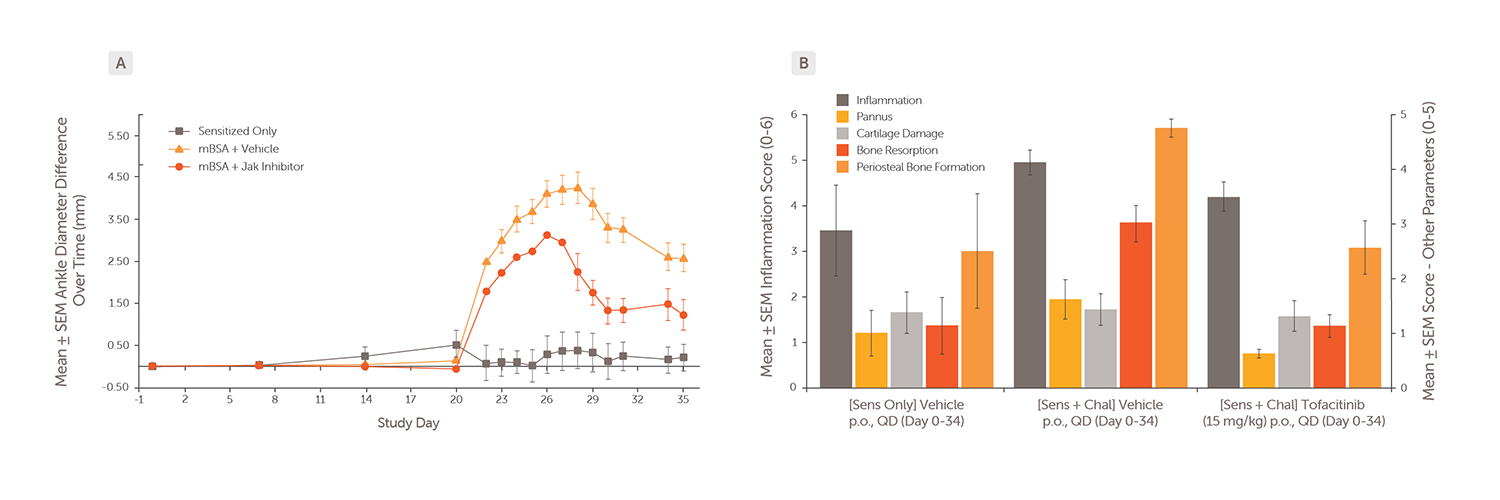
Mice were sensitized with oxazolone by epicutaneous application (Day 1) and challenged by repeated applications to both ears three times per week from Day 11 until termination. Skin samples collected on Day 32 were stained with H&E. (A) Skin section from non-diseased control animal. (B) Skin section from a chronic oxazolone-induced atopic dermatitis mouse showing epidermal hyperplasia (large arrow) and moderate epidermal inflammation with hyperkeratosis (small arrow). D = dermal layer. (C) Histopathology analysis showed epidermal inflammation (dark grey bars), hyperplasia (light grey bars), erosion (orange bars), and dermal inflammation (red bars) in chronic oxazolone-induced mice versus non-diseased controls.
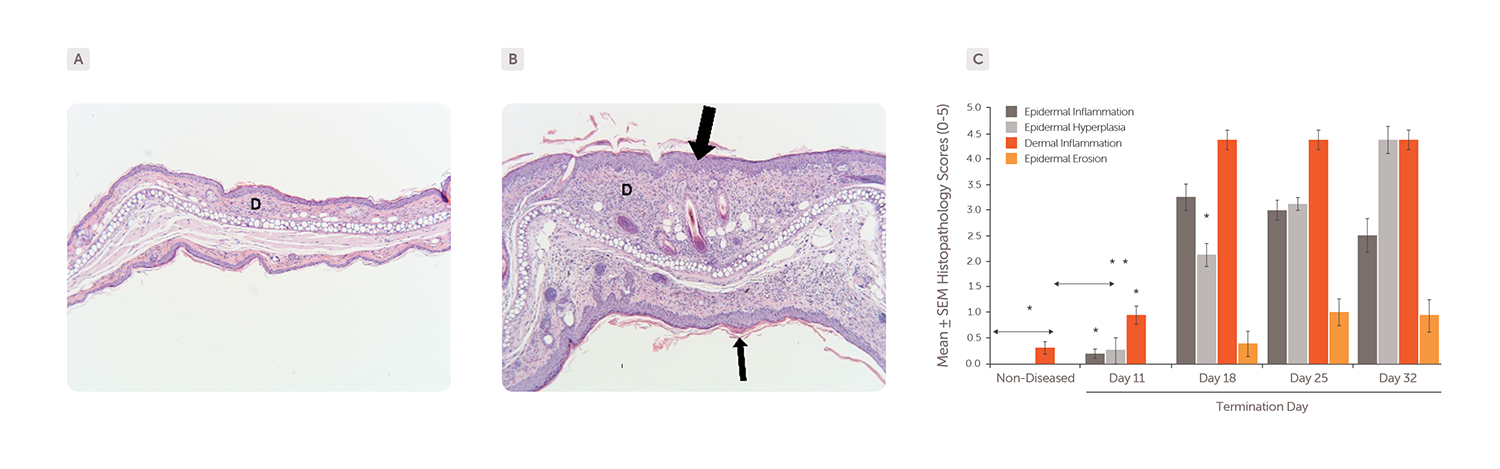
 Atopic dermatitis was induced by daily epicutaneous application of MC903 to one ear for 14 days. Ear thickness was measured daily through Day 15 in MC903-treated mice (grey squares) and vehicle-treated controls (red circles). Data represent mean ± SE ear caliper difference (right minus left).
Atopic dermatitis was induced by daily epicutaneous application of MC903 to one ear for 14 days. Ear thickness was measured daily through Day 15 in MC903-treated mice (grey squares) and vehicle-treated controls (red circles). Data represent mean ± SE ear caliper difference (right minus left).
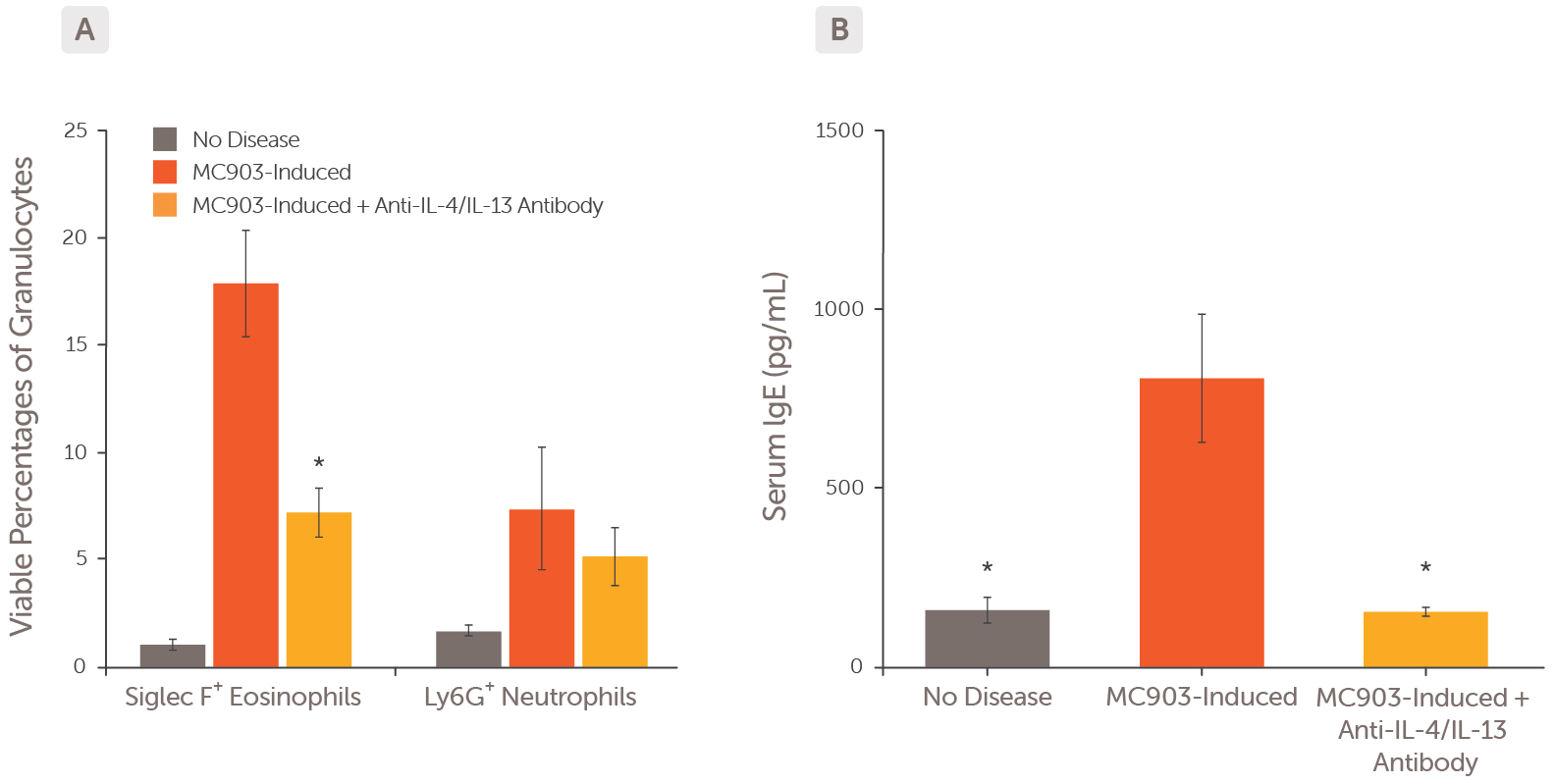 Atopic dermatitis was induced by daily epicutaneous application of MC903 to both ears for 14 days. (A) Eosinophil and neutrophil viability were assessed by flow cytometry in non-diseased controls (grey bars), MC903-induced mice (red bars) and MC903-induced mice treated with an anti-IL-4/IL-13 antibody (orange bars). (B) Serum IgE levels were measured in non-diseased controls (grey bar), MC903-induced mice (red bar) and anti-IL-4/IL-13 antibody-treated mice (orange bar). * p < 0.05 vs MC903-induced mice.
Atopic dermatitis was induced by daily epicutaneous application of MC903 to both ears for 14 days. (A) Eosinophil and neutrophil viability were assessed by flow cytometry in non-diseased controls (grey bars), MC903-induced mice (red bars) and MC903-induced mice treated with an anti-IL-4/IL-13 antibody (orange bars). (B) Serum IgE levels were measured in non-diseased controls (grey bar), MC903-induced mice (red bar) and anti-IL-4/IL-13 antibody-treated mice (orange bar). * p < 0.05 vs MC903-induced mice.
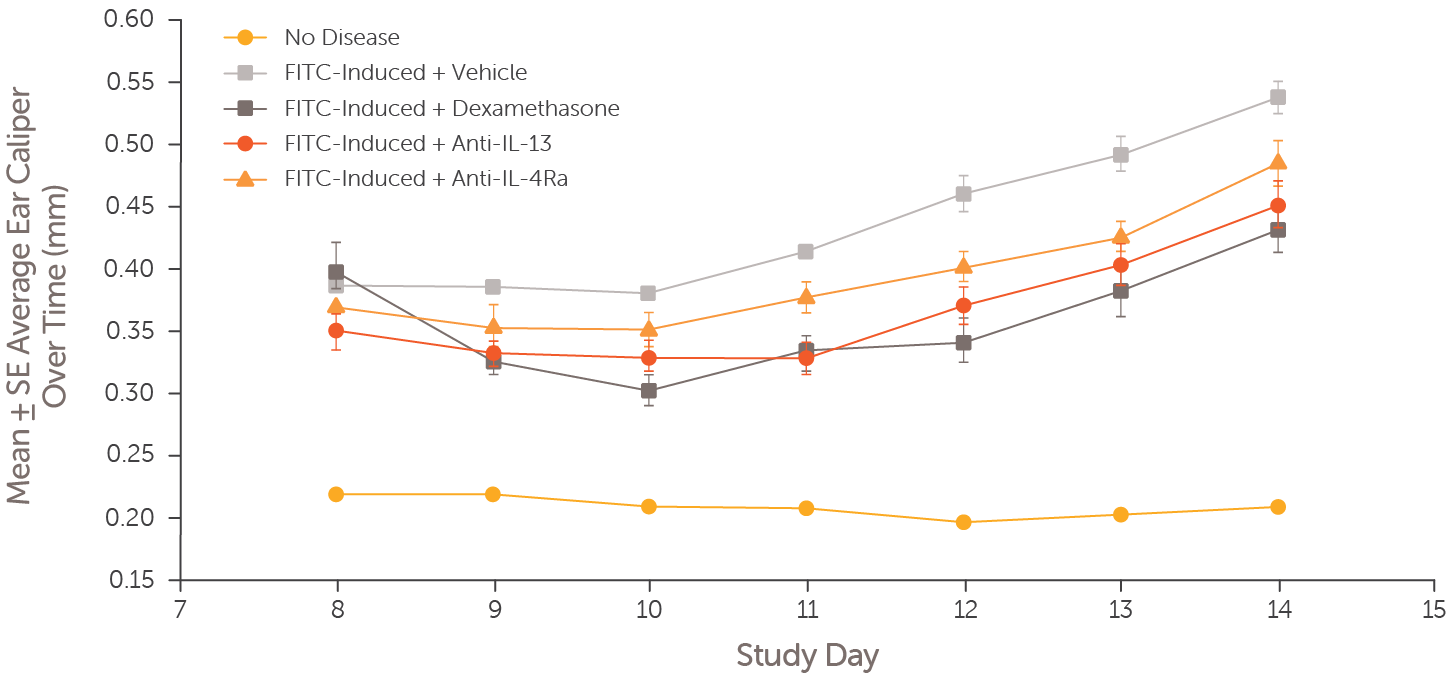 Atopic dermatitis was induced in mice by epicutaneous FITC application on Days 0-1, followed by challenge applications to both ears on Days 7-13. Ear thickness was measured daily through Day 14 in atopic dermatitis mouse models treated with dexamethasone (Days 8-13; dark grey boxes), an anti-IL-13 antibody (Day 8; red circles), anti-IL-4Ra antibody (Day 8; orange triangles), vehicle (Days 8-13; light grey boxes), and non-diseased controls (orange circles).
Atopic dermatitis was induced in mice by epicutaneous FITC application on Days 0-1, followed by challenge applications to both ears on Days 7-13. Ear thickness was measured daily through Day 14 in atopic dermatitis mouse models treated with dexamethasone (Days 8-13; dark grey boxes), an anti-IL-13 antibody (Day 8; red circles), anti-IL-4Ra antibody (Day 8; orange triangles), vehicle (Days 8-13; light grey boxes), and non-diseased controls (orange circles).
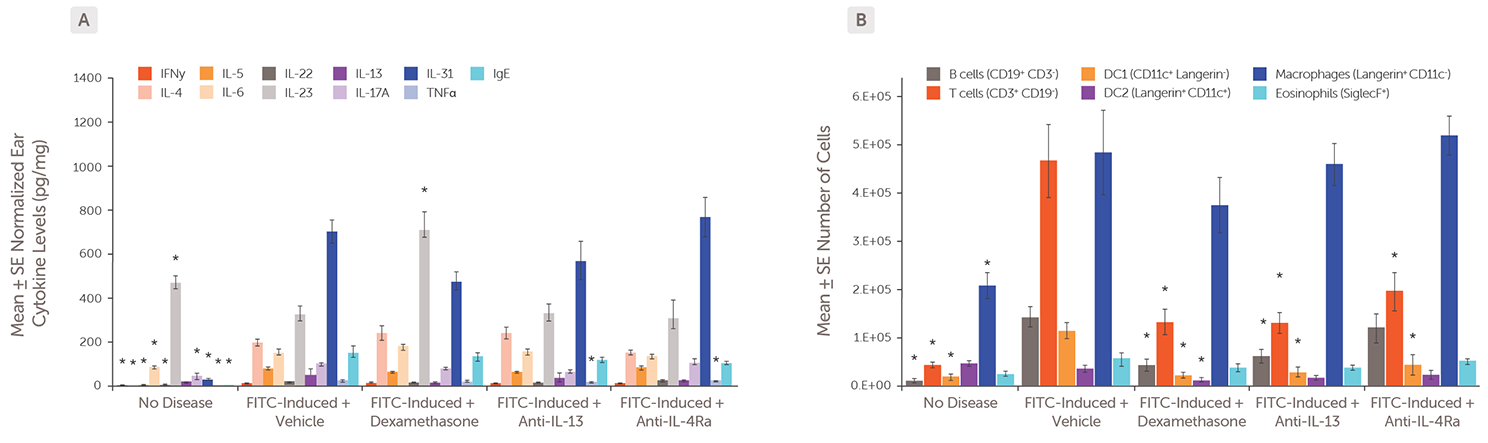
Atopic dermatitis was induced by epicutaneous FITC application on Days 0–1, followed by challenge applications to both ears on Days 7–13. Ear tissue was collected on Day 14 for analysis of (A) cytokine levels and (B) immune cell populations in FITC-induced atopic dermatitis models treated with dexamethasone (Days 8-13), an anti-IL-13 antibody (Day 8), anti-IL-4Ra antibody (Day 8), vehicle (Days 8-13), or non-diseased control animals. * p < 0.05 vs FITC-Induced + vehicle model.
PSORIASIS
Psoriasis is a chronic, immune-mediated inflammatory skin disease characterized by well-demarcated, scaly plaques, most commonly affecting the scalp, elbows, knees, and trunk. It is primarily driven by the IL-23/Th17 axis, with elevated levels of IL-23, IL-17A, IL-22, and TNF-α promoting keratinocyte hyperproliferation, neutrophil infiltration, and persistent skin inflammation. In more severe or treatment-resistant cases, CD4+ Th1 cells and IFN-γ may also contribute to disease progression. We offer validated in vivo psoriasis models that closely recapitulate key features of the IL-23/Th17-driven immune response.
Key Models
- IL-23-Induced Psoriasis Model
- Imiquimod (IMQ)-Induced Psoriasis Model
Example Data
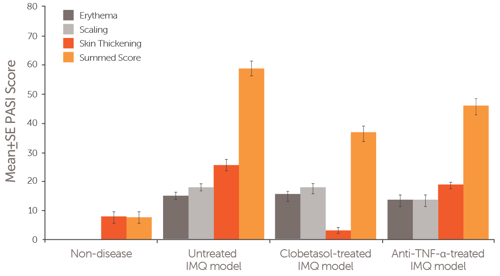
Psoriasis was induced in female Balb/c mice by IMQ application to the shaved and depilated dorsal region of the mouse. During a 7-day study, mice were treated with either Clobetasol (days 0-6), an anti-TNF-α antibody (days 0 and 3), or remained untreated. The severity and extent of psoriasis plaques were assessed using the Psoriasis Area and Severity Index (PASI) measure. The erythema (dark grey bars), scaling (light grey bars), and skin thickening (red bars) of the plaques, as well as the summed score (orange bars), were compared between the IMQ-induced psoriasis models and non-disease mice.
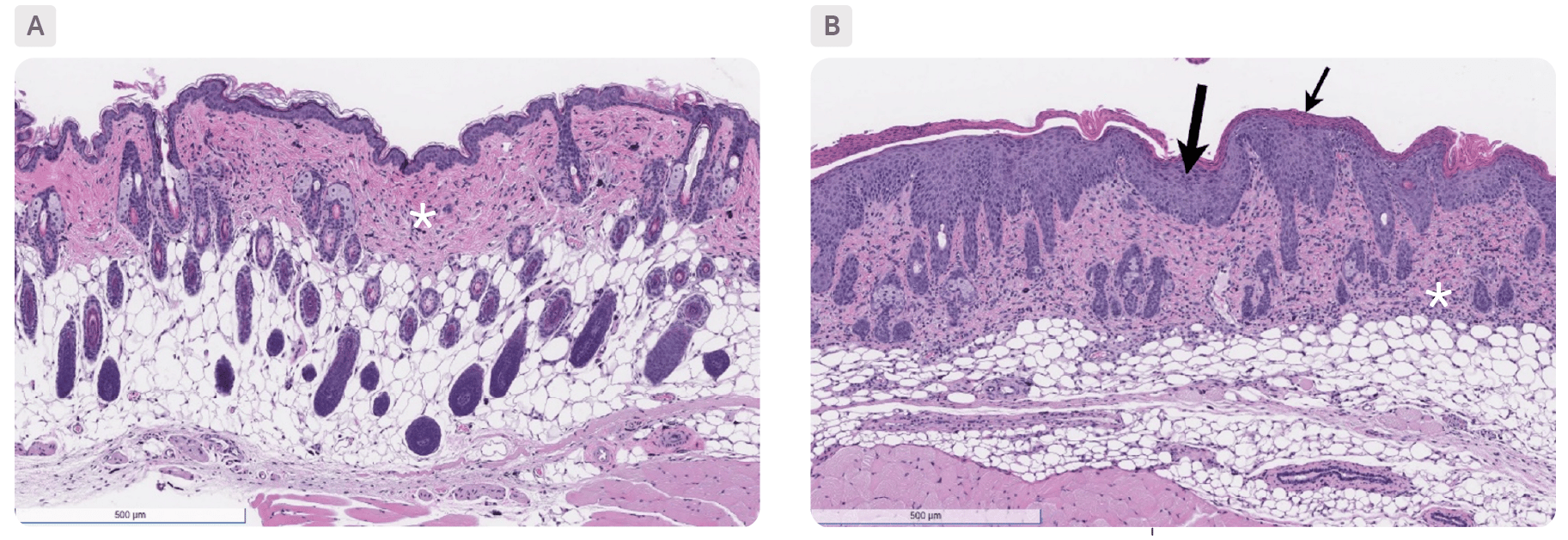
Skin sections, stained with H&E, from a non-disease control animal (A) and an IMQ-induced psoriasis mouse model (B). The control animal shows very minimal dermal inflammation (*) while the IMQ-induced model shows marked epidermal inflammation (small arrow), severe dermal inflammation (*) and moderate epidermal hyperplasia (large arrow).
IRRITANT CONTACT INFLAMMATION
Contact dermatitis is an inflammatory skin condition triggered by exposure to external irritants, resulting in localized erythema, swelling, and pruritus. Irritant contact dermatitis results from direct damage to the skin barrier and activation of innate immune pathways. It is characterized by the release of proinflammatory cytokines such as IL-1 and TNF-α, as well as reactive oxygen species. We offer a validated in vivo model of irritant contact dermatitis induced by the topical application of phorbol ester 12-O-tetradecanoylphorbol-13-acetate (TPA), enabling reproducible evaluation of barrier disruption and innate immune activation.
Key Models
- TPA-Induced Contact Inflammation Mouse Model (12-O-tetradecanoylphorbol-13-acetate)
- Plasma Extravasation
Not seeing the model you need? Contact us to discuss developing a new skin inflammation model for your drug development program.
Clinical Assessment of Inflammatory Skin Disease Models
We conduct a broad range of clinical assessments to evaluate disease severity and treatment response in our inflammatory skin disease models. These endpoints are critical for evaluating the activity of a therapeutic agent. Common endpoints include:
- Visual scoring (erythema, scaling, swelling, skin thickening)
- Ear or skin thickness measurements
- Lesion scoring (epidermal hyperplasia, erosion, dermal inflammation)
- Body weight tracking
- Cytokine and chemokine analysis (local and systemic)
- Flow cytometry for immune cell profiling (FACS)
- IgE quantification
- Caliper measurements (e.g., ankle swelling in DTH models)
Additional endpoints and model-specific assays can be incorporated based on your compound’s mechanism of action and study objectives. Our team collaborates closely with scientists to customize study designs for maximum translational value.
Histopathological Analysis
Histological evaluation is a core component of our preclinical dermatology studies. Tissue sections are assessed by ACVP board-certified pathologists to characterize inflammation, immune infiltration, epidermal remodeling, edema, and other disease-relevant features. These data support efficacy claims, dose optimization, and mechanistic insights for dermatologic drug candidates.
Learn more about our histology services.
Integrated Study Design and Customization
Inotiv offers flexible, client-focused study designs that integrate in vivo, in vitro, and ex vivo methods to support dermatology drug discovery. Studies can be tailored to assess efficacy, safety, and immune modulation using:
- PK/PD blood collections
- Cytokine/chemokine analysis (ELISA, Luminex®)
- CBC/clinical chemistry
- Immunohistochemistry
- Soft tissue collection for downstream assays
We partner with clients to ensure study endpoints align with therapeutic goals, helping generate translatable data that de-risk early-stage programs.